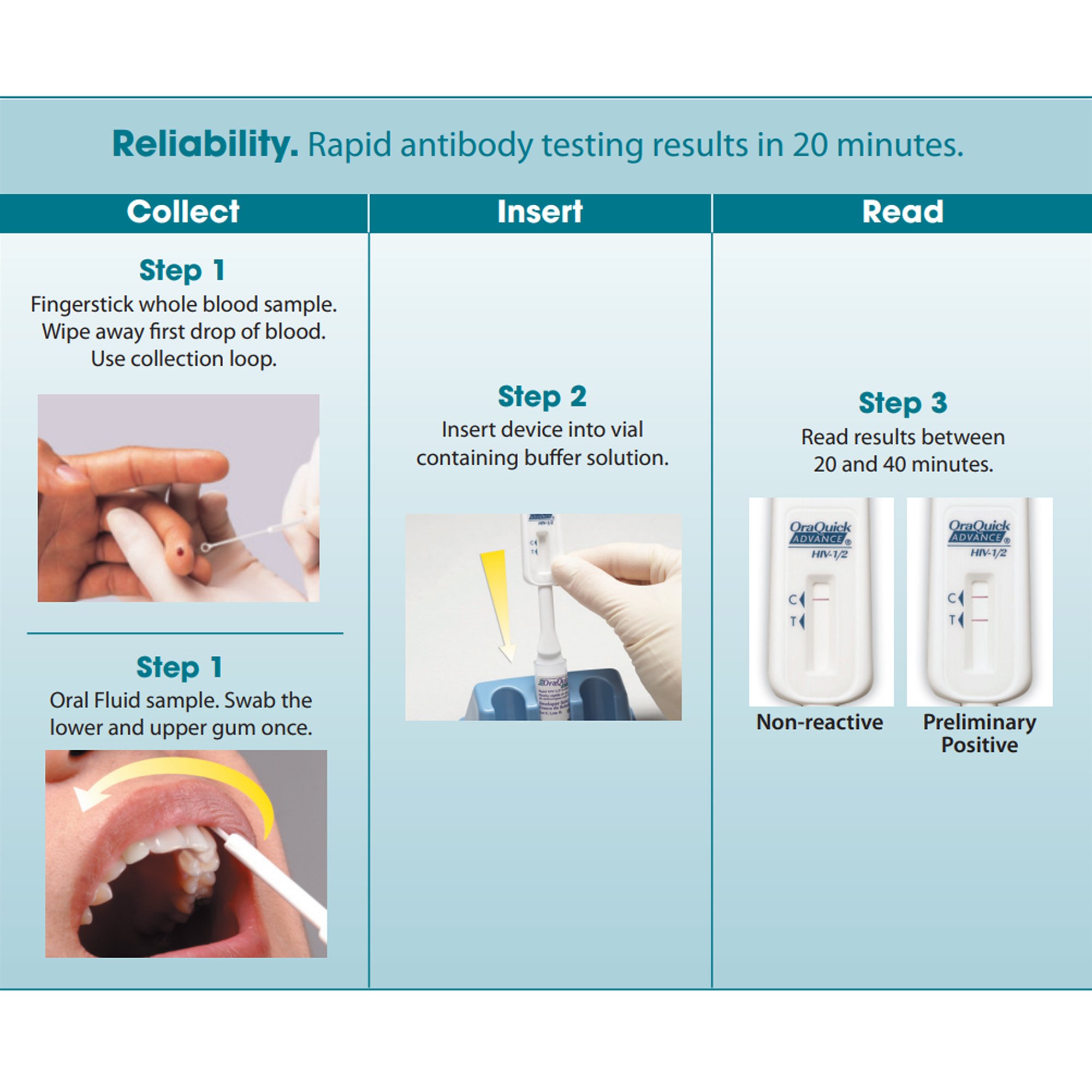
-
- Ambulatory Equipment
- Apparel
- Appliances (Durable Goods)
- Body Pressure Relief and Positioning
- Clinical Laboratory
- Diagnostic Instruments and Supplies
- Drainage and Suction
- Furnishings
- Gloves
- Housekeeping
- Implants
- Incontinence
- Indicators and Signage
- Instruments
- IV Therapy
- Needles and Syringes
- Nurse's Station and Office Supplies
- Nutritionals and Feeding Supplies
- Orthopedic
- Ostomy
- other
- Personal Hygiene
- Pharmaceuticals
- Physical Therapy
- Respiratory
- Sterilization
- Textiles
- Training and Education
- Urological
- Utensils
- Wound Care
- Wound Closure